
BIOBANKING
Collections
BIOBANKING
Collections
The Biobank Côte d’Azur collects human samples associated with various organs and pathologies.
For each of these, we collect numerous biosamples such as tissue fragments (lesional and peri-lesional tissues), biofluids and derived bioproducts. Each sample is associated with the patients’ data, the sample preparation methods and the different tests that are performed in our laboratory.
Thoracic collection
/ COVID-19 collection
The thoracic collection is the largest in the Biobank Côte d’Azur, includin [...]
See more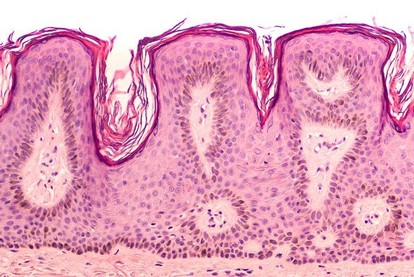
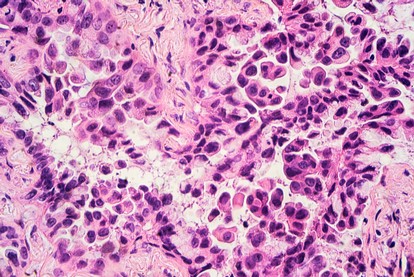
Dermatological collection
The dermatological collection essentially consists of melanoma specimens. M [...]
See more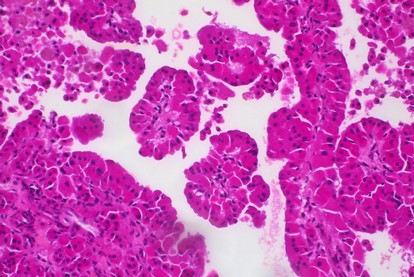
Thyroid COLLECTION
The thyroid collection is the oldest established in the Biobank Côte [...]
See more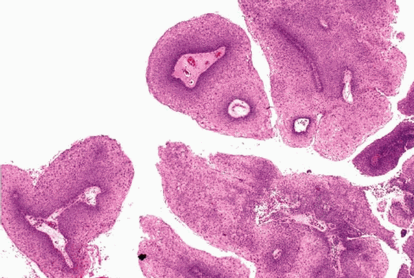
Head and neck Collection
The head and neck collection includes upper aerodigestive tract (UAT) and n [...]
See more
Lymphoma Collection
The lymphoma collection includes all type of lymphoma from several location [...]
See more
The Ophtalmology Collection
The ophthalmology collection includes specimens from orbital, ocular and co [...]
See moreAbout
Our Collections
The flagship collections of the Biobank Côte d’Azur are focused on thoracic, dermatological and thyroid pathologies. Other collections relating to ENT, lymphoma and ophtalmological organs has also been established. Most of the stored tissues relate to tumors, but many also concern non-neoplastic diseases, inflammatory pathologies and infectious diseases.
One of the strengths of the Biobank Côte d’Azur is the collection of both tumor tissue and adjacent non-tumor tissue for the same patient.
In addition to tissue sampling, blood and urine can be collected, usually immediately before surgery.
Different categories of collections are present in our biobank: tissues, liquids, nucleic acids and cells.
The tissue collections are preserved in different forms: frozen tissue is stored at -80°C, whereas tissue that is formaldehyde fixed and paraffin-embedded is stored at room temperature or at 4°C.
In addition to the tissue collections, collections of biological fluids are also preserved (blood and derivatives, urine). Other derived products are also prepared from the samples: constitutional DNA (extracted from whole blood), RNA and DNA from the tumor and plasma circulating tumor DNA.
Finally, cell collections have been established: primary thyroid epithelial cells and peripheral blood mononuclear cells.
For each sample included in the biobank, the clinical data (personal and family history, exposure factors, biology, radiology, treatments and clinical follow-up) and pathological (macroscopy and histology) are collected and recorded in different formats (databases, computer files, paper archiving). In addition to these clinical annotations, the informed consent signed by the patient is included for each sample.
As soon as the biobank was created, a prospective consensual collection policy was established in close partnership with the corresponding surgeons and clinicians for the targeted collections. A retrospective collection of consents, for those patients whose signatures were not requested preoperatively, was put in place to increase the acquisition rate associated with the samples collected for research purposes.
The collections are made available to private or public partners for research purposes. The mission of the Biobank Côte d’Azur is also to be involved in national clinical research projects. The biobank is actively engaged in several National projects such as the AIR-COPD-CTC, Melbase, Cryostem, Ensemble and OFSEP projects.